Barry Bruce and colleagues shine new light on the mechanisms of photosynthesis.
Professor Barry Bruce, Associate Professor Rajan Lamichhane, both of the Department of Biochemistry and Cellular and Molecular Biology (BCMB), and colleagues co-authored a paper that newly reveals the dynamics of photosynthesis at the cellular level.
The team, which included graduate student Sree Kavya Penneru—who Bruce and Lamichhane co-mentor—and post-doctoral researcher Sriram Tiruvadi-Krishnan, published their findings—”Single-molecule fluorescence and cross-linking reveal ligand-gated Toc34 oligomerization dynamics”—in Biophysical Journal in March.
The study reflects a highly productive collaboration. Bruce has investigated the mechanisms of chloroplast biogenesis for more than three decades, and this work extends that research into a powerful new dimension by introducing single-molecule resolution to the problem, made possible through the involvement of Lamichhane, a world leader in single-molecule FRET methodology. His expertise and custom-built instrumentation at UT Knoxville enabled the sophisticated nature of these experiments, resulting in a major technical and conceptual advance that allows parts of chloroplast biogenesis to be studied with more detail than was previously possible.
Mapping the Gatekeepers of Photosynthesis: How Proteins Find Their Way Inside
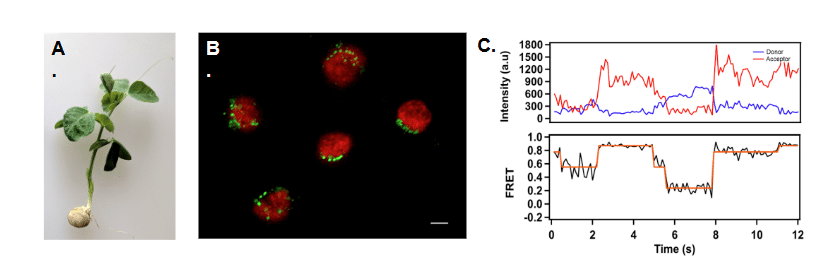
- Image A shows a 13-day old dwarf pea seedling. Image by Sree Kavya Penneru of the Bruce Lab
- Image B shows isolated intact chloroplasts from dwarf pea seedlings, visualized by chlorophyll autofluorescence (red) and the immunolabeling of the Toc34 receptor (green). These Toc34-containing complexes mark locations where thousands of nuclear-encoded proteins are recognized and guided into the organelle after being made in the cell. Image by Praveena Suresh
- The graph in image C shows a representative time-dependent intensity trace of Toc34 labelled with Alexa 555 and Alexa 647 showing Forster Resonance Energy Transfer (FRET) from the team’s recently published work (Biophysical Journal 125, 1475–1493, March 17, 2026).
“This is a unique collaboration that bridges disciplines,” said Bruce. “Lamichhane’s work on GTPases in GPCR systems—central to drug discovery—is now being applied to a different but equally important GTPase system that controls chloroplast function in plants. This shift allows us to use advanced single-molecule tools to study a process that is fundamental to how plants produce the food, fiber, and fuel on which society depends.”
Researchers have previously characterized the structure of the chloroplast, the specialized organelles in plants and algae that are the primary site of photosynthesis. Chloroplasts need to import most of their proteins from outside the organelle, and the Toc34 receptor helps control this entry process. Toc34 works as a pair of identical proteins (a homodimer), which can be thought of as two parts of the same lock on a door. Together, this Toc34 pair helps recognize incoming proteins and decide when the “door” should open.
“The two halves of the lock communicate with each other and use a small energy molecule (GTP) to switch between different states,” said Bruce. “However, we still do not fully understand how this ‘double lock’ changes shape or activity to control protein import.”
The team will extend their strategy to other membrane-bound systems in future studies, expanding their approach to build a more complete model of the dynamic and coordinated events that take place within the workings of photosynthesis.
“In the future, we plan to double the complexity of these experiments by moving beyond the Toc34 homodimer to examine the full two-receptor system involving both Toc34 and Toc159,” said Bruce. “By revealing how the Toc34 ‘lock’ opens and closes, this work creates an opportunity either to make protein import more efficient—supporting stronger plant growth—or to precisely ‘jam the lock,’ offering a plant-specific strategy for new herbicides.”
by Randall Brown

